Northwestern University has developed a small, flexible, implantable device that relieves pain on demand without the use of drugs. It works by providing targeted cooling to block pain signals to the brain. Dimensions are only 5 mm at the widest point the biocompatible device dissolves after it is no longer needed, similar to absorbable sutures.
The researchers said the "first-of-its-kind device could provide a much-needed alternative to opioids and other highly addictive drugs."
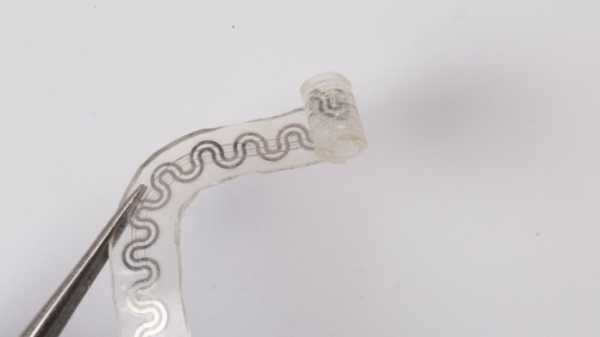
< p id="caption-attachment-232043" class="wp-caption-text">The implantable device consists of two separate microfluidic channels: one for liquid coolant and the other for gas. When mixing in the chamber, the liquid evaporates at a certain point on the nerve to ensure precise cooling. (Source: Northwestern University)
According to the researchers, the water-soluble device "gently" wraps around the nerves to provide precise cooling that numbs the nerves and blocks pain signals to the brain. The cooling effect is caused by two tiny microfluidic channels in the device. One channel contains a liquid coolant (perfluoropentane), and the other channel contains dry nitrogen. When a liquid and a gas combine in a chamber, the reaction causes the liquid to vaporize.
According to the researchers, the liquid coolant is clinically approved as an ultrasound contrast agent and for pressurized inhalers.
<p>"We're specifically targeting the peripheral nerves that connect your brain and spinal cord to the rest of your body," study co-author Dr. Matthew McEwan of Washington University School of Medicine in St. Louis said in a statement. “These are the nerves that transmit sensory stimuli, including pain. By providing a cooling effect to only one or two target nerves, we can effectively modulate pain signals in one specific part of the body.”
The device also contains a built-in sensor that "monitors the temperature of the nerve." to make sure it doesn't get too cold, which could cause tissue damage," the researchers said.
"Excessive cooling can damage the nerve and the fragile tissues around it," Northwestern's John A. Rogers, who led the device's development, said in a statement. "Therefore, it is necessary to precisely control the duration and temperature of cooling. By monitoring the temperature on the nerve, the flow rate can be adjusted automatically to establish a point that blocks pain in a reversible and safe way."
Rogers is the Louis Simpson and Kimberly Querrey Professor of Materials Science and Engineering, Biomedical Engineering, and Neurological Surgery at Northwestern University's McCormick School of Engineering and Feinberg School of Medicine. He is also the founding director of the Querrey Simpson Institute for Bioelectronics.
An external pump is used by the patient to remotely activate the device, as well as to increase or decrease the intensity. When the device is no longer needed, it is naturally absorbed into the body within days or weeks.
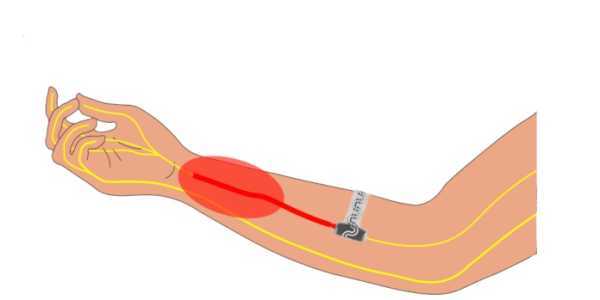
The implanted device wraps around a peripheral nerve to block signals to the brain. The red oval in the picture indicates the area of pain. (Source: Northwestern University)
The researchers said that “the device will be most valuable for patients undergoing routine surgeries or even amputations that typically require post-operative medication. Surgeons could implant the device during the procedure to ease the patient's post-operative pain."
The study was published in the July 1 issue of the journal Science. It describes the design and efficacy in an animal model. The research was supported by the Phil and Penny Knight Campus to Accelerate Scientific Impact, the Querrey Simpson Institute for Bioelectronics, and the National Science Foundation.
This is the third example of bioresorbable electronic devices from Rogers' lab. The concept of transient electronics was introduced in 2012, followed by a demonstration of a bioresorbable electronic device in 2018 and a transient pacemaker in 2021.
Source: electronicproducts.com





